The Instrument Qualification Kits are designed to assist laboratories in meeting the updated requirements of USP chapter 857 and European Pharmacopoeia (EP) 2.2.25 for UV-Visible spectrophotometer validation. These standards, effective since December 2019 and January 2020 respectively, mandate qualification of absorbance linearity, wavelength accuracy, stray light, and resolution/bandwidth using references that bracket the measurement parameters.
The Basic Qualification Kits include carefully selected Certified Reference Materials (CRMs) for qualifying wavelength accuracy, absorbance accuracy and linearity over specified ranges, as well as stray light and resolution. Potassium Chloride solution is included as a sensitive indicator of optical degradation even outside far UV applications. While these kits cover the common pharmaceutical analysis ranges, additional references are available for customized measurement conditions.
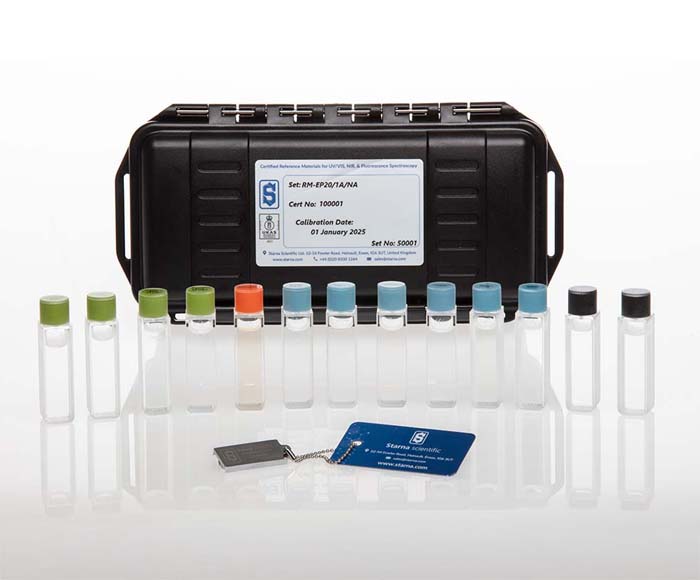
The Instrument Qualification Kits are designed to assist laboratories in meeting the updated requirements of USP chapter 857 and European Pharmacopoeia (EP) 2.2.25 for UV-Visible spectrophotometer validation. These standards, effective since December 2019 and January 2020 respectively, mandate qualification of absorbance linearity, wavelength accuracy, stray light, and resolution/bandwidth using references that bracket the measurement parameters.
The Basic Qualification Kits include carefully selected Certified Reference Materials (CRMs) for qualifying wavelength accuracy, absorbance accuracy and linearity over specified ranges, as well as stray light and resolution. Potassium Chloride solution is included as a sensitive indicator of optical degradation even outside far UV applications. While these kits cover the common pharmaceutical analysis ranges, additional references are available for customized measurement conditions.